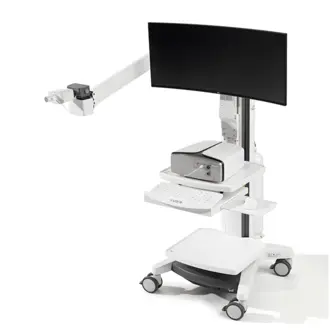
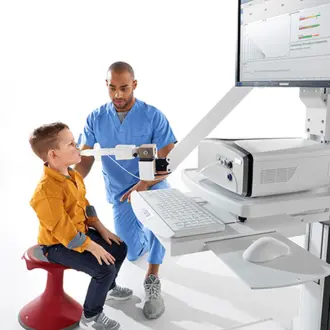
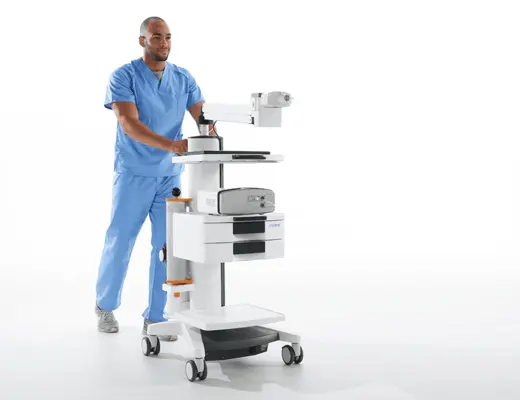
Vyntus™ ONE Pulmonary Function System
Designed to be hassle-free for clinicians and friendly for patients, Vytnus™ ONE is incredibly capable with a modular design for an impressive array of features. Tests are more accurate, less intrusive and less stressful for patients.
Vyntus™ ONE offers complete pulmonary function testing such as spirometry, DLCO, nitrogen washout lung volumes, LCI and MIP/MEP. It can be expanded to do cardiopulmonary stress testing, creating a single device at a fraction of the footprint.
Vyntus™ ONE can be configured to accommodate any medical center's needs. Visit our Knowledge Base and learn how the latest SentrySuite software helps you achieve the best possible results according to the latest ATS/ERS 2019 Spirometry guidelines.
Features & Benefits
- Modular design with 3 configuration options
- Validated, simple hygiene and maintenance routines
- Factory Calibrated, Ultrasonic Flow Sensor with Double Shot Technology
- Meets latest ATS/ERS DLCO guidelines
- Innovative eDemand valve requiring less patient effort
- Tool free O2 cell exchange
- Mobile version for PFT testing outside the lab
Customer Education Resources
Please note that all products, services, or features of products and services may not be available in your local area. Please check with your local Vyaire representative.