Mechanical Ventilation
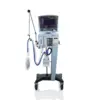
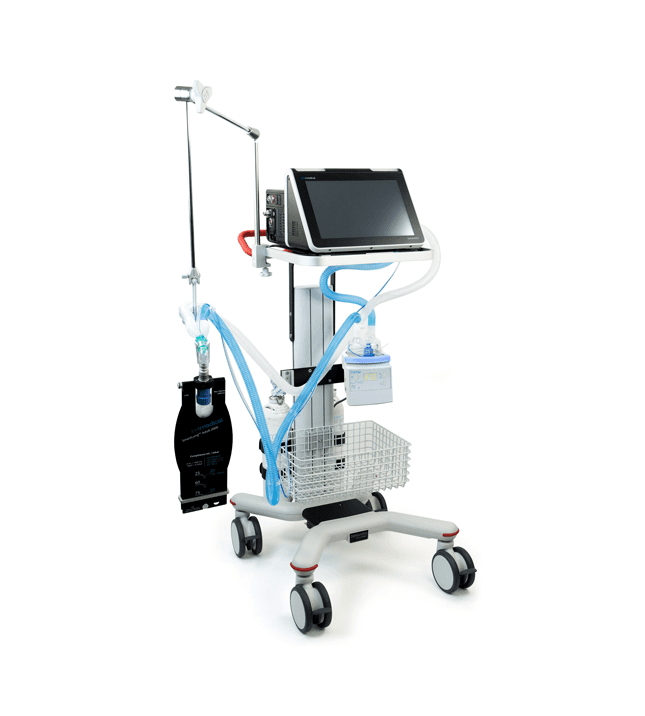
AVEA™ CVS Ventilation System
For clinicians and administrators in the acute care environment, the Avea™ CVS is a comprehensive neonatal through adult ventilation system with the complete package of all advanced features and maneuvers in one device. It helps empower clinicians at the bedside to improve patient outcomes and decrease costs-of-care, while the Respiratory Knowledge Portal receives and processes data, converting it to actionable insights to help clinicians improve respiratory care processes.
Features & Benefits
- Neonatal, pediatric and adult capable
- Invasive and Non-invasive Ventilation (including Infant nCPAP and nIMV )
- Active exhalation valve promotes ventilator synchrony and comfort
- Internal battery provides up to 30 minutes of power
- Internal compressor/ air source (runs on DC/ battery power)
Customer Education Resources
Please note that all products, services, or features of products and services may not be available in your local area. Please check with your local Vyaire representative.
Related Products
The Latest News
Contact Form
Vyaire Medical Completes Business Unit Sales
Vyaire has completed transactions with ZOLL Medical Corporation and Trudell Medical Limited.